Prof. Slavka S. Tcholakova, Ph.D.
Head of the Laboratory for Active Formulations and Materials
Interests
- Formation, stability and rheology of foams and emulsions
- Mechanism of action of antifoams
- Surfactant aggregates and mesophases in bulk: structure, rheology, stability
- Biophysics & colloid science in fat digestion and drug delivery systems
- Defoamers and antifoams
- Physicochemical theoretical models for foamability, emulsification; foam rheology
- Natural based surfactants
Bio
Prof. Slavka Tcholakova received M.Sc. in Chemistry (1996), Ph.D. in Physical Chemistry (2004), Assistant Professor (2006), Associate Professor (2009), and became a Full Professor in 2013 in the Faculty of Chemistry and Pharmacy, Sofia University, Bulgaria. She was a head of the Department of Chemical and Pharmaceutical Engineering (DCPE) in Sofia University from 2015 to 2024. Currently, she is a head of the Laboratory “Active formulations and materials” at DCPE. She has been a visiting researcher in the Research Center Paul Pascal, CNRS, Bordeaux, France (1997).
Her research interests include formation and stability of emulsions; rheology of foams and emulsions; protein adsorption in relation to emulsion stability; foam stability in the presence of antifoams; in-vitro models for digestion and bioavailability of hydrophobic molecules and the respective experimental methods. So far, she has published 150 research and review papers, cited over 6300 times (h-index = 44). She has been leading over 70 projects and participating in more than 30 projects with international companies (BASF, Unilever, Saint Gobain, Wacker, Lubrizol, PepsiCo, Altana, BYK Chemie, Productolysa, etc.).
She has been a supervisor and co-supervisor of 16 completed PhD Theses. She was the recipient of the “Best Young Scientist” award for 2006 of the Sofia University Foundation “St. Kliment Ohridski”. In 2018, Prof. Tcholakova received the National Bulgarian award “Pythagoras” for high scientific achievements in category Natural Sciences.
Based on her publications and citations in 2023 and 2024, Prof. Tcholakova achieved high rankings in the Chemical Physics category of the Stanford/Elsevier’s Top 2% Scientists Ranking: In 2024, she was ranked 1712 out of 106,831 scientists; In 2025, she was ranked 1764 out of 115,551 scientists.
Publications
Most recent publications
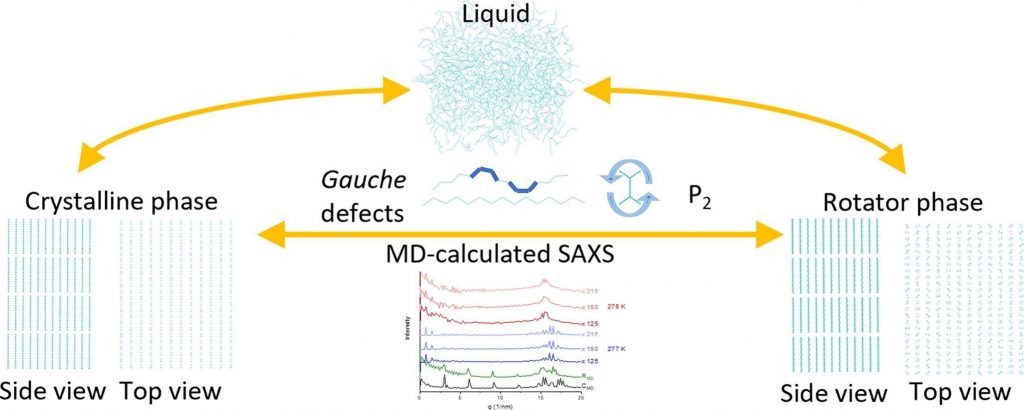
Computational freezing of pentadecane
Molecular dynamics simulations are employed to investigate the crystallization of pentadecane-containing systems. Reference crystalline and rotator phase constructed from crystallographic data benchmark the structures formed upon cooling. Phase identification is achieved through global and local structural descriptors, with the fraction of gauche conformations and angular P₂ profiles outlined as the most sensitive indicators.
Pentadecane exhibits a markedly more stable rotator phase than hexadecane. This is confirmed by simulations spanning more than 30 K for the reference rotator phase. In this temperature range, a model regular rotator phase of pentadecane remains stable without undergoing significant structural changes, showing high reproducibility across independent trajectories. This contrasts hexadecane, for which a rotator phase rapidly transforms toward a triclinic structure [Iliev et al. 2023]. The presence of a surfactant in the system promotes heterogeneous nucleation, shifts crystallization to higher temperatures, and stabilizes the rotator phase, in agreement with experiments.
A computationally efficient protocol for simulating SAXS spectra from MD trajectories is proposed. The simulated spectra align very well with experimental data for both crystalline and rotator phases. Crystallographic lattice parameters can be extracted from the most intense SAXS peaks, even for experimentally unknown structures, demonstrating the general applicability of the approach to solid-state phase analysis.
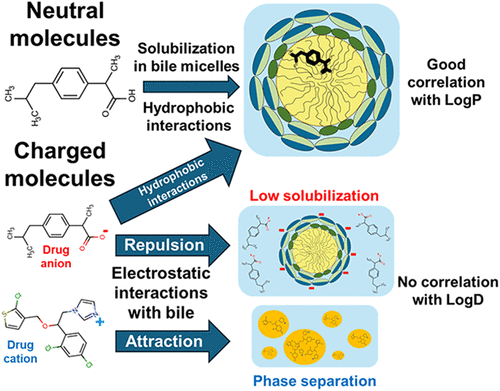
Drug solubilization in simulated intestinal fluids vs lipophilicity: Does charge matter?
Solubilization of poorly water-soluble drugs in human intestinal fluids influences oral absorption and is linked to food effects. Current empirical equations for calculating intestinal solubilization via lipophilicity are built on limited data and do not adequately account for drug ionization. We aim to expand the data set and build a model to clarify the link between lipophilicity and solubilization for charged compounds. We determined the aqueous solubility, octanol–water partition coefficient, and solubilization in fed-state simulated intestinal fluids (FeSSIF) of 26 hydrophobic drugs. Combined with literature data, a good correlation (R2 = 0.74, n = 198) between intestinal solubilization and LogP/D was observed. However, data segregation showed that the solubilization of neutral compounds correlated very well with LogP (R2 = 0.89, n = 114), whereas the correlation with LogD was lost for the charged compounds (R2 = 0.40, n = 84). To better understand this behavior, the pH of FeSSIF was varied to study the solubilization of the same compounds in the neutral and charged states. While a very good correlation between solubilization and LogD was observed in the neutral state of the compounds (R2 = 0.92, n = 8), the correlation was again lost (R2 = 0.02, n = 4) in their charged state. Electrostatic interactions were suggested to play a key role in the unexpectedly low solubilization of anionic drugs and in the phase separation observed for cationic drugs. The presented insights further advance the understanding of the solubilization of hydrophobic drugs in biorelevant media and provide a foundation for broader and improved modeling approaches.
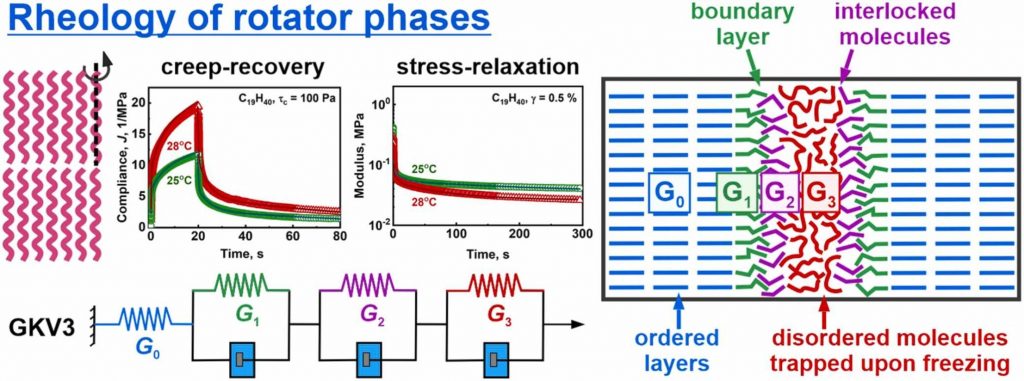
Rheological characterization of solid lipids with domain structure
Phase behavior of lipids is of primary importance for the manufacturing and applications of foods, cosmetics and pharmaceuticals, as well as for the functions of biological membranes. Upon cooling, the molten bulk lipids crystallize into ordered domains which determine their rheological properties. While storage and loss moduli are typically used to describe these properties, their direct connection to the underlying molecular rearrangement remains poorly understood. In the current study, we performed a detailed rheological characterization of the rotator phases (intermediate phases between fully ordered crystalline and completely disordered liquid phases) formed in bulk linear alkanes. Large series of stress-relaxation and creep-recovery experiments were performed and interpreted, using generalized Kelvin-Voigt model with one spring, connected in series with three combined elements of a spring and a dashpot. We determined the elasticities and viscosities of all these rheological elements, along with the respective three relaxation times of the combined elements. These relaxation times are governed by different molecular processes in the sheared samples and differ by three orders of magnitude: t1 ≈ 0.45 s and t2 ≈ 8–9 s are related to local molecular rearrangements at the domain boundaries, while t3 ≈ 140–200 s most probably describes the rearrangement of disordered lipid molecules entrapped between the ordered domains. The storage and loss moduli, calculated from the constants of the generalized Kelvin-Voigt model, were in a very good agreement with those measured directly in amplitude sweep and temperature ramps oscillatory tests, thus supporting the self-consistency of data interpretation. The methodology presented here is applicable to other polycrystalline lipid materials with 2D or 3D domain structures, providing a valuable framework for interpreting their rheological behavior.
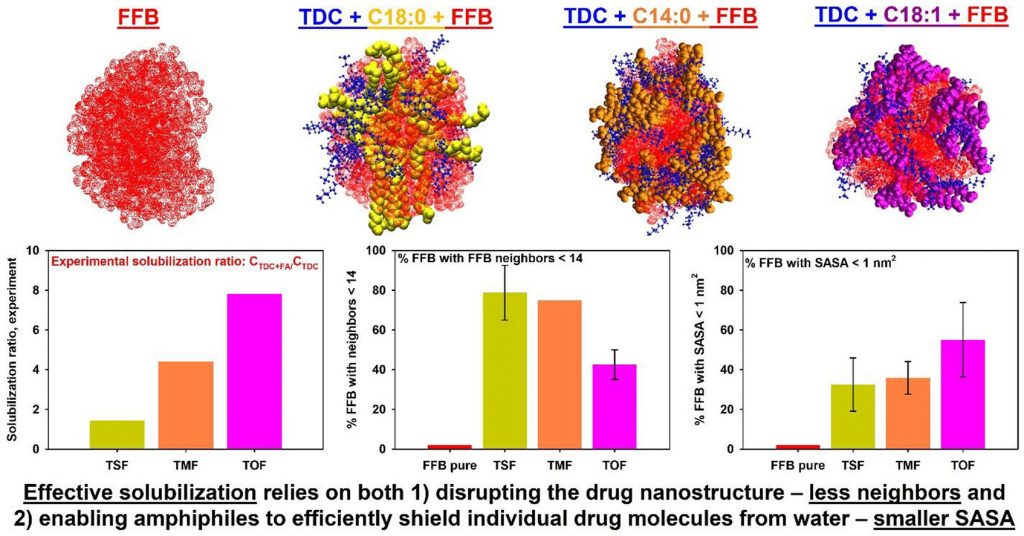
Computational modeling of the early-stage solubilization of a fenofibrate aggregate into mixed bile salt and fatty acids micelles
The behavior of a fenofibrate aggregate in water and its interaction with pure sodium taurodeoxycholate (TDC) micelles, as well as with mixed micelles of TDC and fatty acids (myristic, oleic, and stearic), was studied using atomistic molecular dynamics simulations. For each system, 500 ns trajectories were generated and analyzed in terms of the association of amphiphiles around the drug aggregate and their impact on the morphology and intermolecular interactions of the drug molecules. The results show that in the absence of fatty acids TDC covers the surface of the drug aggregate, shielding it from water without disrupting its structure. In contrast, all studied fatty acids intercalate between the drug molecules in the aggregate. However, myristic and stearic acids do not shield sufficiently the drug molecules from water, while oleic acid significantly reduces the contact between water and drug molecules. Two key requirements for effective solubilization are identified: (1) the ability of amphiphiles to disrupt the nanostructure of the drug aggregate, and (2) the flexibility and capability of amphiphiles to reduce interactions between water and hydrophobic drug molecules. These requirements were used to evaluate the efficiency of fatty acids in increasing the solubilization capacity of TDC micelles. A very good correlation was established between the efficiency determined from molecular dynamics simulations and experimental data known from the literature. The methodology developed in this study could be widely used to compare the efficiency of new lipid-based drug delivery formulations to solubilize hydrophobic drug molecules.
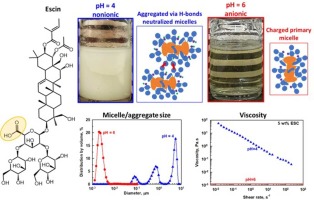
Escin solutions: Effects of pH and electrolytes on their behavior
Escin is a triterpenoid saponin with one carboxyl group which is non-ionized at pH 4.7. The major aim of the current study is to determine how the electrolyte concentration affects the properties of concentrated escin solutions (5 wt% and 10 wt%) at pHs of 4, 6, and 8. Ionized escin molecules at pH >4.7 form charged micelles that repel one another when there is no added electrolyte and solutions remain clear and stable for more than a month. Lowering the pH to 4 leads to formation of uncharged micelles. These micelles attract each other and form inter-micellar hydrogen bonds, which enable formation of micrometer aggregates that cause turbidity and phase separation. The addition of background electrolytes to the solutions at pHs of 6 and 8 screens the electrostatic repulsion between micelles, causing partial aggregation of the micelles and gelation of solutions. As the salt concentration increases, the viscosity of the escin solution also increases, reaching a maximum—similar to the behavior observed with conventional surfactants. However, the mechanism behind this viscosity maximum is different. In solutions of conventional surfactants, the maximum is due to the formation of worm-like micelles, whereas the maximum for escin solutions is due to formation of a network of escin aggregates that imparts yield stress and elasticity to the solution. These dispersions remain stable for at least one month at room temperature and can be used as cosmetic and detergent formulations.