Drug solubilization in simulated intestinal fluids vs lipophilicity: Does charge matter?
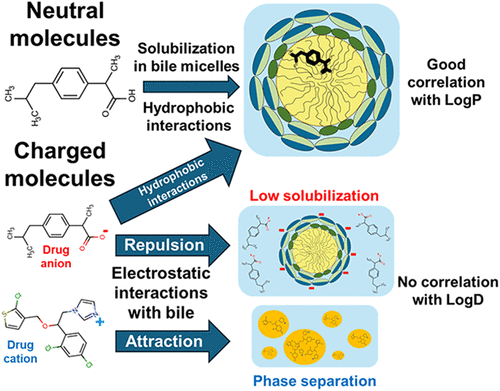
Solubilization of poorly water-soluble drugs in human intestinal fluids influences oral absorption and is linked to food effects. Current empirical equations for calculating intestinal solubilization via lipophilicity are built on limited data and do not adequately account for drug ionization. We aim to expand the data set and build a model to clarify the link between lipophilicity and solubilization for charged compounds. We determined the aqueous solubility, octanol–water partition coefficient, and solubilization in fed-state simulated intestinal fluids (FeSSIF) of 26 hydrophobic drugs. Combined with literature data, a good correlation (R2 = 0.74, n = 198) between intestinal solubilization and LogP/D was observed. However, data segregation showed that the solubilization of neutral compounds correlated very well with LogP (R2 = 0.89, n = 114), whereas the correlation with LogD was lost for the charged compounds (R2 = 0.40, n = 84). To better understand this behavior, the pH of FeSSIF was varied to study the solubilization of the same compounds in the neutral and charged states. While a very good correlation between solubilization and LogD was observed in the neutral state of the compounds (R2 = 0.92, n = 8), the correlation was again lost (R2 = 0.02, n = 4) in their charged state. Electrostatic interactions were suggested to play a key role in the unexpectedly low solubilization of anionic drugs and in the phase separation observed for cationic drugs. The presented insights further advance the understanding of the solubilization of hydrophobic drugs in biorelevant media and provide a foundation for broader and improved modeling approaches.

