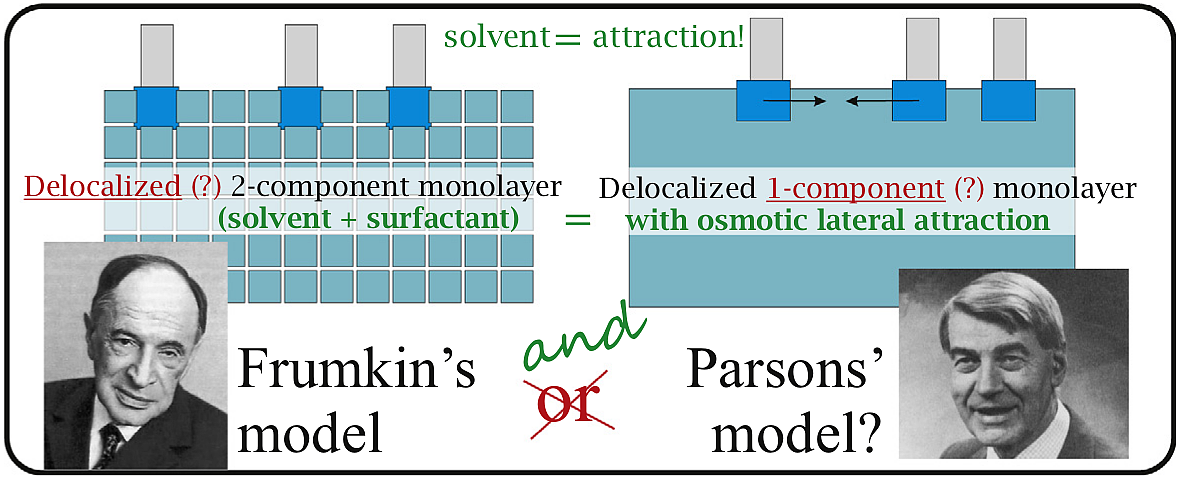
A molecular thermodynamic model is derived for an uncharged delocalized surfactant monolayer adsorbed at a liquid interface, taking explicit account for the solvent molecules present in the monolayer. The model is based on the scaled particle theory of hard-disc mixtures, and is also extended to sticky discs (i.e. attraction between the adsorbed molecules). Upon compression of the adsorbed layer, the solvent is expelled from it. The respective osmotic effect on the equation of state is shown to be equivalent to an effective lateral depletion attraction between the surfactant molecules. This effective osmotic cohesion causes an increase of the value of the attraction parameter b of the monolayer. The smaller the size of the surfactant polar head group is, the larger the effective attraction the model predicts. This trend is verified with data for the adsorption at water|air surface of alcohols, undissociated acids, and hexaethylenglycol monoalkyl ethers. The proposed theory allows the amount of solvent in the monolayer to be estimated, which is shown to be important for the neutron reflectivity of the surface.