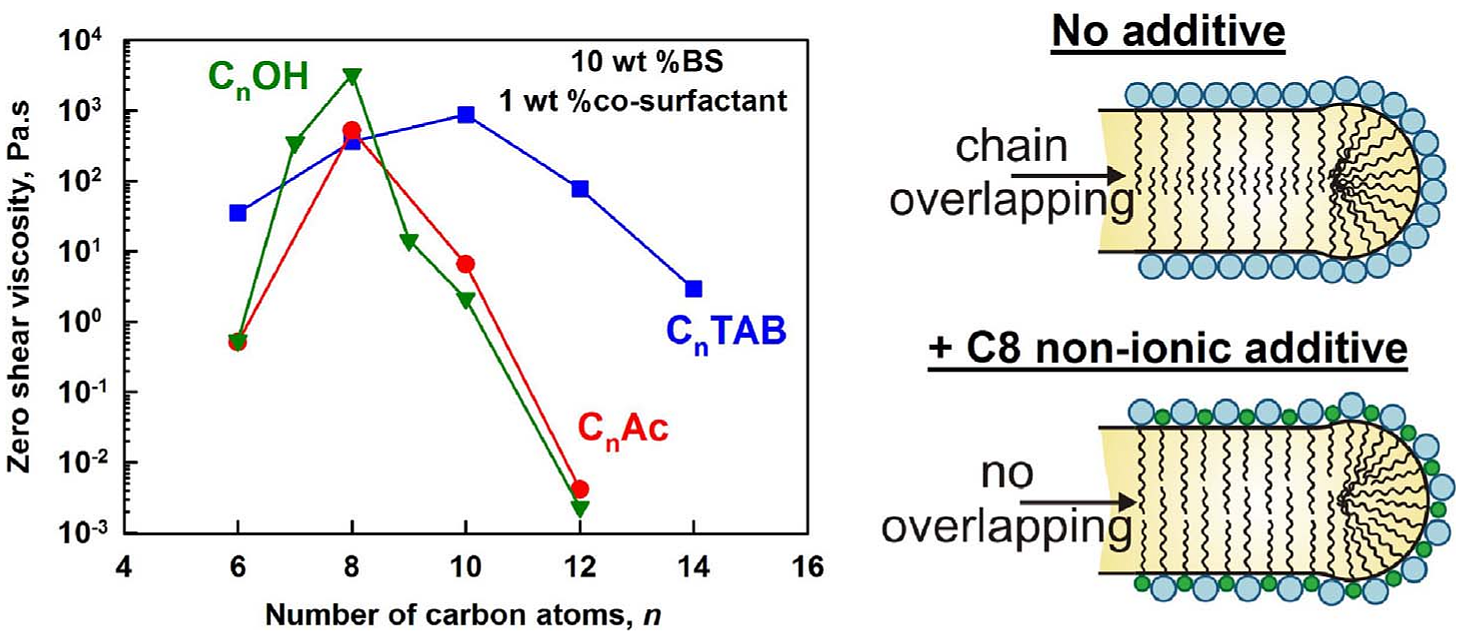
The rheological properties of surfactant solutions play an important role in their applications. Here we study systematically how different cosurfactants affect the rheological properties of mixed solutions of the anionic surfactant sodium laurylether sulfate (SLES) and the zwitterionic cocoamidopropyl betaine (CAPB). Mixed SLES + CAPB solutions are used in various formulations due to their excellent foaming and cleaning properties. These solutions possess nearly Newtonian behavior and low viscosity. However, the addition of cosurfactants in low concentrations may significantly increase the viscosity of these solutions and even transform them into viscoelastic gels. Here we study systematically a wide range of ionic and nonionic cosurfactants with different head-groups, chain-lengths and structures of the hydrophobic tails (linear, branched, double bonded). We reveal an optimal chain-length of 8–10 carbon atoms of the cosurfactant molecules which ensures the highest viscoelasticity of the triple SLES + CAPB + cosurfactant solutions. Cationic cosurfactants and nonionic ones with small head-group (e.g. fatty alcohols and fatty acids) are most efficient in increasing the solution viscoelasticity. There is a hierarchy in the parameters of the cosurfactant molecules which govern this behavior − most important is the head-group charge, then the chain-length and, finally, the presence of branching and double bonds in the hydrophobic tails. The observed trends are explained with the effect of the cosurfactant molecules on the properties of the entangled wormlike micelles formed in these solutions.