Reexamining the enhanced solubility of sodium laurate/sodium oleate eutectic mixtures
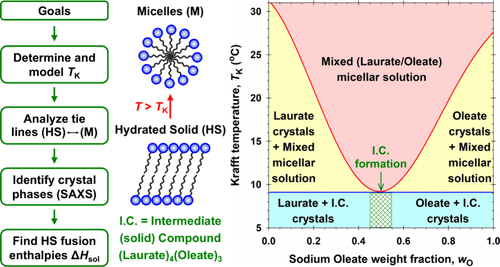
Mixtures of multiple surfactants that have superior performance to the individual components are highly sought-after commercially. Mixtures with a reduced Krafft point (TK) are particularly useful as they enable applications at lower temperatures. Such an example is the soap maker’s eutectic: the mixture of sodium laurate (NaL) and sodium oleate (NaOl). A true eutectic implies that the two surfactants do not mix in the solid state but mix readily in the micellar solution above TK, leading to a sharp TK depression at a specific composition. However, the NaL/NaOl mixture shows a broad TK depression of >15 °C at a NaOl weight fraction (wO) of about 0.5. Our tie-line analysis shows that pure NaL and NaOl do not coexist in the solid phase on either side of the TK minimum. X-ray analysis of the isolated solids with varying wO reveals that a unique intermediate compound (I.C.) forms in the solid state with a NaL-to-NaOl mole ratio of about 4/3. Below the TK minimum, NaL and the I.C. coexist in the solids for wO 0.5. Each pair of solids exhibits eutectic or monotectic solubility behavior, and the congruent I.C. melting point is so close to that of the eutectic point(s) that a broad TK minimum ensues. Thermal analysis and modeling via the freezing-point depression approach support the above interpretation. The fact that surfactants with other headgroups but the same blend of chain lengths do not exhibit similar depressed TK is a topic for further study.

