Alkyl sucrose esters vs. Brijs: How chain length and temperature impact surface and foam properties
The primary objective of this study is to determine the similarities and differences in the surface, film, and foam properties of alkyl sucrose esters (SEs) with high monoester content (≥ 70 %) and polyoxyethylene alkyl ethers (Brijs) with a high number of ethoxy groups (≥ 20) in their head group. Experiments were conducted using surfactant molecules with alkyl chain lengths of 12, 16, and 18 carbon atoms at concentrations between 0.01 and 1 wt%, within a temperature range of 25 °C to 60 °C.
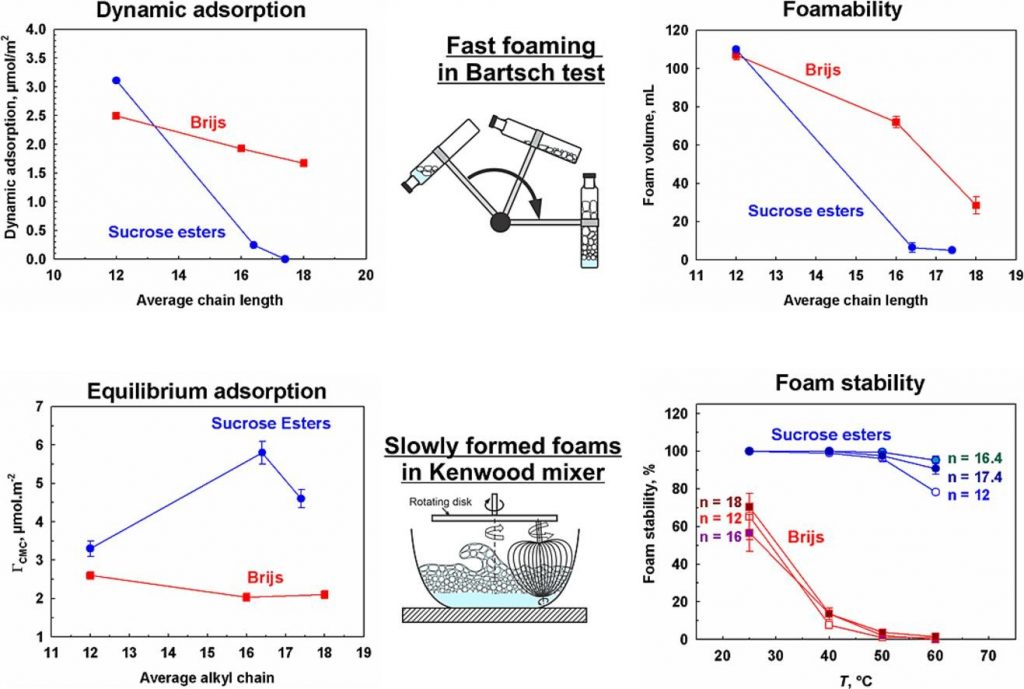
The lag time for surfactant adsorption increased with surfactant chain length, decreased with temperature, and significantly decreased with surfactant concentration for both types of studied surfactants. However, increasing the chain length from 12 to 18 carbon atoms led to a 10-fold increase in lag time for Brijs and more than a 600-fold increase for SEs. This effect rendered longer-chain SEs incapable of forming voluminous foam in Bartsch test and led to pronounced coalescence between the bubbles after their separation from the sparger in the foam rise method, resulting in foams with very large bubbles, which exhibited lower stability. The utilization of a Kenwood mixer for foam generation provided sufficient time for longer-chain SE molecules to adsorb on the bubble surfaces and to produce voluminous foams with small bubbles, which remained stable even at 60 °C. In contrast, foams generated from Brijs solutions are very unstable at 60 °C. The long-standing stability of SEs foam was attributed to the formation of mixed mono- and diesters adsorption layers on the bubble surfaces.

