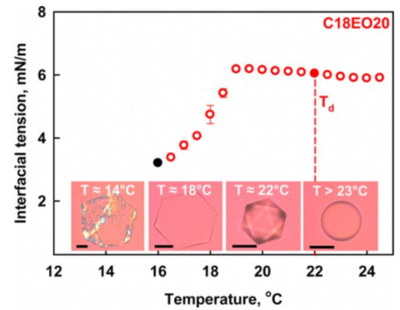
 Two recent studies (Denkov et al., Nature 528 (2015) 392 and Guttman et al., PNAS 113 (2016) 493) demonstrated that micrometer sized n-alkane drops, dispersed in aqueous surfactant solutions, can break their spherical symmetry upon cooling and "self-shape" into a variety of regular shapes, such as fluid polyhedra, platelet-shaped hexagons, triangles, rhomboids, toroids, and sub-micron in diameter fibers. In the first study, the observed phenomenon was explained by a mechanism involving the formation of interfacial multilayer of self-assembled alkane molecules in the so-called “rotator phases”, templated by the frozen surfactant adsorption layer. Such phases are known to form in alkane droplets under similar conditions and are sufficiently strong to deform the droplets against the capillary pressure of a finite interfacial tension of several mN/m. The authors of the second study proposed a different explanation, namely, that the oil-water interfacial tension becomes ultra-low upon cooling, which allows for surface extension and drop deformation at negligible energy penalty. To reveal which of these mechanisms is operative, we measure in the current study the temperature dependence of the interfacial tensions of several systems, undergoing such drop shape transitions. Our results unambiguously show that the drop “self-shaping” is not related to ultra-low oil-water interfacial tension, as proposed by Guttmann et al. These results support the mechanism proposed by Denkov et al. which implies that the large bending moment, required to deform an oil-water interface with interfacial tension of 5 to 10 mN/m, is generated by interfacial multilayer of selfassembled alkane molecules.
Two recent studies (Denkov et al., Nature 528 (2015) 392 and Guttman et al., PNAS 113 (2016) 493) demonstrated that micrometer sized n-alkane drops, dispersed in aqueous surfactant solutions, can break their spherical symmetry upon cooling and "self-shape" into a variety of regular shapes, such as fluid polyhedra, platelet-shaped hexagons, triangles, rhomboids, toroids, and sub-micron in diameter fibers. In the first study, the observed phenomenon was explained by a mechanism involving the formation of interfacial multilayer of self-assembled alkane molecules in the so-called “rotator phases”, templated by the frozen surfactant adsorption layer. Such phases are known to form in alkane droplets under similar conditions and are sufficiently strong to deform the droplets against the capillary pressure of a finite interfacial tension of several mN/m. The authors of the second study proposed a different explanation, namely, that the oil-water interfacial tension becomes ultra-low upon cooling, which allows for surface extension and drop deformation at negligible energy penalty. To reveal which of these mechanisms is operative, we measure in the current study the temperature dependence of the interfacial tensions of several systems, undergoing such drop shape transitions. Our results unambiguously show that the drop “self-shaping” is not related to ultra-low oil-water interfacial tension, as proposed by Guttmann et al. These results support the mechanism proposed by Denkov et al. which implies that the large bending moment, required to deform an oil-water interface with interfacial tension of 5 to 10 mN/m, is generated by interfacial multilayer of selfassembled alkane molecules.